Answer : The correct answer for theoretical yield = 2.73 g
Theoretical yield : It is defined as maximum amount of yield that is expected to be produce from reactants . The theoretical yield is decided by amount of product formed by Limiting Reagent .
Theoretical yield can be calculated bu using stroichiometric relation between mole of limiting reactant and product .
The reaction of Benzil can be written as :
one mole of Benzoin produces one mole of Benzil ( Image ) .
Since only one reactant , Benzoin is present so limiting reactant can be considered as Benzoin .
Theoretical yield can be calculates in following steps :
Step 1 : Conversion of mass of Benzoin to its mole .
Mass can be converted to mole using following formula :
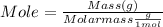
Molar mass of Benzoin (C₁₄H₁₂O₂ ) = 212.24
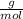
Plugging value of Mass and molar in mole formula :
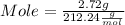
Mole = 0.013 mol
Step 2 : To find mole ratio of Benzil to Benzoin .
Mole ratio can be find out from balanced reaction . From balanced reaction one mole of Benzoin giving one mole of Benzil , Hence mole ratio = 1 : 1
Step 3 : Find mole of Benzil
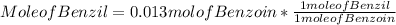
Mole of Benzil = 0.013 mol
Step 4 : To find mass of Benzil
Molar mass of Benzil (C₁₄H₁₀O₂ ) = 210.232
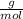
Plugging value in mole formula
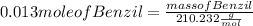
Multiplying both side by 210.232
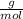
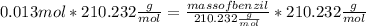
Mass of Benzil = 2.73 g
Hence Theoretical mass of Benzil = 2.73 g