Answer:
2 L Ne
General Formulas and Concepts:
Math
Pre-Algebra
Order of Operations: BPEMDAS
- Brackets
- Parenthesis
- Exponents
- Multiplication
- Division
- Addition
- Subtraction
Chemistry
Atomic Structure
- Using Dimensional Analysis
- STP (Standard Conditions for Temperature and Pressure) = 22.4 L per mole at 1 atm, 273 K
Step-by-step explanation:
Step 1: Define
0.07 mol Ne (g)
Step 2: Identify Conversions
STP - 22.4 L per mole
Step 3: Convert
- Set up:
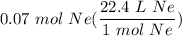
- Multiply:
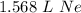
Step 4: Check
Follow sig fig rules and round. We are given 1 sig fig.
1.568 L Ne ≈ 2 L Ne