Answer:
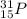 is the isotopic notation of the atom
is the isotopic notation of the atom
Step-by-step explanation:
The isotope notation is:
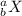
Where a is the mass number = Number of protons + Number of neutrons
b is atomic number = Number of protons
The atomic number define the nature of the atom, the element with atomic number = 15 is phosphorus, P:
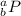
a = 15 protons + 16 neutrons = 31
b = 15
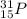 is the isotopic notation of the atom.
is the isotopic notation of the atom.