Answer:
See explanation.
Step-by-step explanation:
Hello!
In this case, since we are asked to note down the complete molecular equation, complete ionic equation and the net ionic equation for the reaction between acetic acid (weak acid) and barium hydroxide (strong base), we proceed as shown below:
- Complete molecular equation:
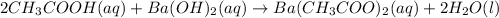
- Complete ionic equation: in this case, since acetic acid is a weak one, it is not ionized, so we do this:
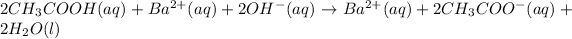
- Net ionic equation: in this case, we cancel out the barium ions as they are the spectators one because they are present at both reactants and products:
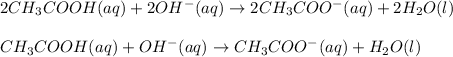
Best regards!