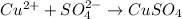Answer:
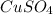
Step-by-step explanation: For formation of a neutral ionic compound, the charges on cation and anion must be balanced. The cation is formed by loss of electrons by metals and anions are formed by gain of electrons by non metals.
Here Copper is having an oxidation state of +2 and sulfate is a polyatomic ion with oxidation state of -2. Thus they combine to form neutral
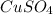 .
.
The cations and anions being oppositely charged attract each other through strong coloumbic forces and form an ionic bond.