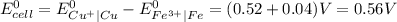Answer:
Standard cell potential is 0.56V
Step-by-step explanation:
Standard reduction potential (
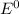 ) are given below-
) are given below-
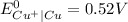
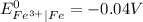
As
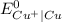 is greater than
is greater than
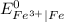 therefore Fe will be oxidized to
therefore Fe will be oxidized to
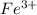 and
and
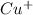 will be reduced to Cu
will be reduced to Cu
Oxidation:
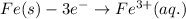
Reduction:
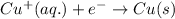
------------------------------------------------------------------------------------------------------
Overall:
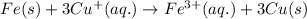
Standard cell potential,