Answer:
Benzoic acid
Step-by-step explanation:
In this case we have a Grignard reaction in which we go from a formula of C6H5Br to C7H6O2. In other words, we add 1 carbon to the molecule. The only way to add one carbon to this molecule is using
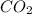 as a reagent. When this compound is used as electrophile a carboxylic acid is produced.
as a reagent. When this compound is used as electrophile a carboxylic acid is produced.
Respect to the NMR info, we have a downfield singlet signal at 12.1 ppm which is characteristic of the acid hydrogens in the carboxylic acids (see figure 1). When a carboxylic group is added to the benzene we will obtain the benzoic acid which fits with the formula and the NMR info given.