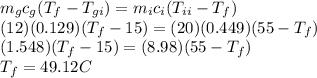Answer:
49.12 °C
Step-by-step explanation:
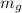 = mass of gold sheet = 12 g
= mass of gold sheet = 12 g
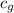 = specific heat of gold = 0.129 J/g°C
= specific heat of gold = 0.129 J/g°C
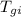 = initial temperature of gold sheet = 15 °C
= initial temperature of gold sheet = 15 °C
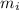 = mass of iron sheet = 20 g
= mass of iron sheet = 20 g
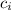 = specific heat of iron = 0.449 J/g°C
= specific heat of iron = 0.449 J/g°C
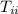 = initial temperature of iron sheet = 55 °C
= initial temperature of iron sheet = 55 °C
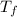 = Final equilibrium temperature
= Final equilibrium temperature
Using conservation of heat