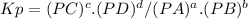Answer:
Kp = 67
Step-by-step explanation:
The reaction is given as:
PCl3 (g) + Cl2 (g) → PCl5 (g)
Now, the Kp expression at equilibrium for this reaction is written as:
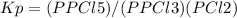
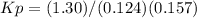
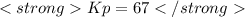
Notes:
- Kp is the gas constant at equilibrium in terms of partial pressures of gas.
- For a reaction: aA + bB <===> cC +dD, the expression of Kp can be written as: