Answer: 386.0 g/mol
Step-by-step explanation:
As the relative lowering of vapor pressure is directly proportional to the amount of dissolved solute.
The formula for relative lowering of vapor pressure will be,
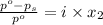
where,
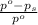 = relative lowering in vapor pressure
= relative lowering in vapor pressure
i = Van'T Hoff factor = 1 (for non electrolytes)
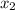 = mole fraction of solute =
= mole fraction of solute =
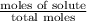
Given : 21.47 g of compound X is present in 233.8 g of diethyl ether
moles of solute (X) =
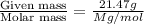
moles of solvent (diethyl ether) =
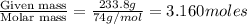
 = mole fraction of solute =
= mole fraction of solute =
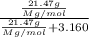
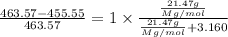
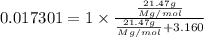
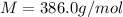
The molecular weight of this compound is 386.0 g/mol