Answer:
-1
Step-by-step explanation:
The relation between Kp and Kc is given below:
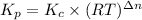
Where,
Kp is the pressure equilibrium constant
Kc is the molar equilibrium constant
R is gas constant , 0.082057 L atm.mol⁻¹K⁻¹
T is the temperature in Kelvins
Δn = (No. of moles of gaseous products)-(No. of moles of gaseous reactants)
For the first equilibrium reaction:
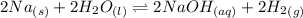
Δn = (No. of moles of gaseous products)-(No. of moles of gaseous reactants) = (2+1)-(2+2) = -1