Answer:
The value of n is 14.
Step-by-step explanation:
To calculate the concentration of solute, we use the equation for osmotic pressure, which is:
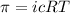
where,
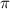 = osmotic pressure of the solution = 57.1 Torr =
= osmotic pressure of the solution = 57.1 Torr =
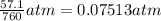
1 atm = 760 Torr
i = Van't hoff factor = 2 (electrolytes)
c = concentration of solute = ?
R = Gas constant =
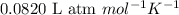
T = temperature of the solution =
![25^oC=[273.15+25]=298.15 K](https://img.qammunity.org/2020/formulas/chemistry/high-school/1vjnzmjlo43s073441qytu4jvdecwaj4kb.png)
Putting values in above equation, we get:
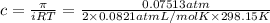
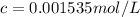
Assuming that molality and molarity in such a dilute solution.
c = m (Molality)
The salt is soluble in water to the extent of 0.036 g per 100 g of water at 25°C
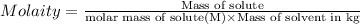
Molality of the solution = m = 0.001535 mol/L
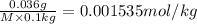
M = 234.53 g/mol
Molar mass of
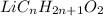 : M
: M
M =
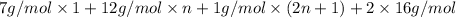
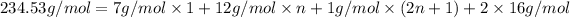
n = 14
The value of n is 14.