Answer:
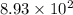 years
years
Step-by-step explanation:
Given that:
Half life = 5730 years
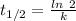
Where, k is rate constant
So,
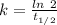
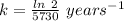
The rate constant, k = 0.00012 years⁻¹
Initial concentration [A₀] = 59 Bq
Final concentration
![[A_t]](https://img.qammunity.org/2020/formulas/chemistry/college/wbj92t0z4axifcyqa24z3ary269op2iva8.png) = 53 Bq
= 53 Bq
Time = ?
Using integrated rate law for first order kinetics as:
![[A_t]=[A_0]e^(-kt)](https://img.qammunity.org/2020/formulas/chemistry/college/wgh5hifj7f12vitsa51kophgqrxxcfit2c.png)
Applying values, we get that:-
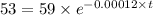
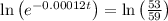
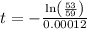
t =
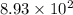 years
years
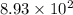 years is the age of the artifact.
years is the age of the artifact.