Answer:
K remains the same;
Q < K;
The reaction must run in the forward direction to reestablish the equilibrium;
The concentration of
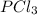 will decrease.
will decrease.
Step-by-step explanation:
In this problem, we're adding an excess of a reactant, chlorine gas, to a system that is already at equilibrium. According to the principle of Le Chatelier, when a system at equilibrium is disturbed, the equilibrium shifts toward the side of the equilibrium that minimizes the disturbance.
Since we'll have an excess of chlorine, the system will try to reduce that excess by shifting the equilibrium to the right. Therefore, the reaction must run in the forward direction to reestablish the equilibrium.
The value of K remains the same, as it's only temperature-dependent, while the value of Q will be lower than K, that is, Q < K, as Q < K is the case when reaction proceeds to the right.
As a result, since
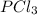 is also a reactant, its concentration will decrease.
is also a reactant, its concentration will decrease.