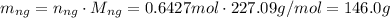Answer:
a.
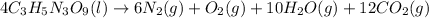
b. 146.0 g
Step-by-step explanation:
Question 1 (a). Just as the problem states, liquid nitroglycerin decomposes into nitrogen gas
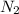 , oxygen gas
, oxygen gas
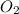 , water vapor
, water vapor
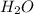 and carbon dioxide
and carbon dioxide
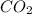 . Let's write the decomposition of nitroglycerin into these 4 components:
. Let's write the decomposition of nitroglycerin into these 4 components:
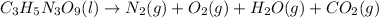
Now we need to balance the equation. Firstly, notice we have 3 carbon atoms on the left and 1 on the right, so let's multiply carbon dioxide by 3:
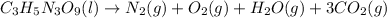
Now, we have 3 nitrogen atoms on the left and 2 on the right, so let's multiply nitrogen on the right by
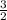 :
:
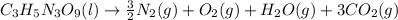
We have 5 hydrogen atoms on the left, 2 on the right, so let's multiply the right-hand side by
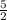 :
:
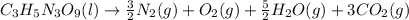
Finally, count the oxygen atoms. We have a total of 9 on the left. On the right we have (excluding oxygen molecule):
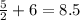
This leaves
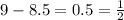 of oxygen. Since oxygen is diatomic, we need to take one fourth of it to get one half in total:
of oxygen. Since oxygen is diatomic, we need to take one fourth of it to get one half in total:
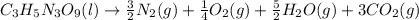
To make it look neater without fractional coefficients, multiply both sides by 4:
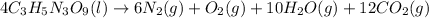
Question 2 (b). Now we can make use of the balanced chemical equation and apply it for the context of this separate problem. We're given the following variables:
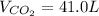
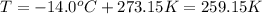
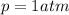
Firstly, we may find moles of carbon dioxide produced using the ideal gas law
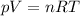 .
.
Rearranging for moles, that is, dividing both sides by RT (here R is the ideal gas law constant):
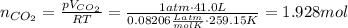
According to the stoichiometry of the balanced chemical equation:
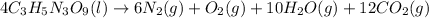
4 moles of nitroglycerin (ng) produce 12 moles of carbon dioxide. From here we can find moles o nitroglycerin knowing that:
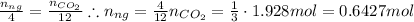
Multiplying the number of moles of nitroglycerin by its molar mass will yield the mass of nitroglycerin decomposed: