Answer:
8.6
Step-by-step explanation:
Using the expression :
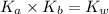
Where,
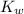 is the dissociation constant of water.
is the dissociation constant of water.
At
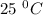 ,
,
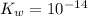
Thus, for benzoic acid , pKa = 4.20
Thus,
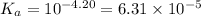
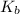 for Sodium benzoate can be calculated as:
for Sodium benzoate can be calculated as:
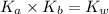
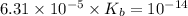
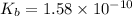
The benzoate ion will dissociate as:-
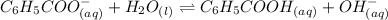
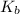 expression is:-
expression is:-
![K_(b)=\frac {\left [ C_6H_5COOH^(+) \right ]\left [ {OH}^- \right ]}{[C_6H_5COO^-]}](https://img.qammunity.org/2020/formulas/chemistry/college/vpnyqo2igt1dm1t43h1yyvvfewu1s7eg38.png)
Given that:-
Moles = 0.100 moles
Volume = 1.00 L
Thus, Concentration = 0.100/ 1.00 M = 0.1 M
Considering the ICE table in the image below.
So,
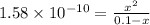
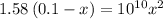
Solving for x, we get that,
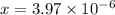
Thus,
![[OH^-]=3.97* 10^(-6)](https://img.qammunity.org/2020/formulas/chemistry/college/56h42xrdcit5gp3yyn8habvls0q0ttjd2l.png)
Also,
![pOH=-log[OH^-]=-log(3.97* 10^(-6))=5.4](https://img.qammunity.org/2020/formulas/chemistry/college/ajkrexn2uxcpgk04yp39eylgytutgwrqvq.png)
Also, pH + pOH = 14
So, pH = 14 - 5.4 = 8.6