Answer:
Balanced equation: 2 [Cr(en)₂(NCS)₂](SCN)(s) → en(g) + [Cr(en)₂(NSC)₂][Cr(en)(NCS)₄](s)
Oxidation states on Cr in the reactant and the products: +3
Step-by-step explanation:
The balanced chemical equation is the following:
2 [Cr(en)₂(NCS)₂](SCN)(s) → en(g) + [Cr(en)₂(NSC)₂][Cr(en)(NCS)₄](s) (1)
In the balanced reaction (1) we have that 2 moles of [Cr(en)₂(NCS)₂](SCN) decompose in 1 mol of ethylenediamine and 1 mol of Cr(en)₂(NSC)₂][Cr(en)(NCS)₄.
Determination of oxidation state of the Cr:
In the reactant of the balanced equation (1), we know that the charge on SCN is -1, so for the complex [Cr(en)₂(NCS)₂](SCN) to be neutral, the charge on the Cr(en)₂(NCS)₂ must be +1, hence the state oxidation on the Cr is:
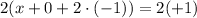
where x: is the oxidation state on Cr, 0: is the oxidation state on ethylenediamine since is neutral, and (+1): is the oxidation state on [Cr(en)₂(NCS)₂].
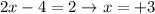
So, the oxidation state of Cr in the reactant is +3.
In the products of the balanced equation (1), the charge on the two complex ions must be (+1) for [Cr(en)₂(NSC)₂] and (-1) for [Cr(en)(NCS)₄] since total charge on the complex [Cr(en)₂(NSC)₂][Cr(en)(NCS)₄] is zero (is neutral), so we have:
![[x + 2(0) + 2(-1)] + [x + 0 + 4(-1)] = 0](https://img.qammunity.org/2020/formulas/chemistry/high-school/7ofa4ctcib9qanxv4qmuufc7hfx3bbmhke.png)
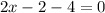
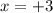
Therefore, the oxidation state on Cr is +3 in the two complex ions in the products.
I hope it helps you!