Answer:
The volume occupied by given molecules of carbon dioxide at STP is 55.86 L.
Step-by-step explanation:
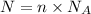
Where:
N = Number of particles / atoms/ molecules
n = Number of moles
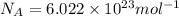 = Avogadro's number
= Avogadro's number
We have:
N =
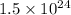
n =?
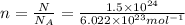
n = 2.4909 moles
Using ideal gas equation:
PV = nRT
where,
P = Pressure of carbon dioxide gas =
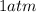 (at STP)
(at STP)
V = Volume of carbon dioxide gas = ?
n = number of moles of carbon dioxide gas =2.4909 mol
R = Gas constant = 0.0821 L.atm/mol.K
T = Temperature of carbon dioxide gas = 273.15 K (at STP)
Putting values in above equation, we get:
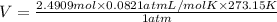
V = 55.86 L
The volume occupied by given molecules of carbon dioxide at STP is 55.86 L.