Answer:
(a) ΔP=0.0245 kPa
(b) P=9.14 kPa
(c)ΔP=0.0245 kPa
Step-by-step explanation:
(a) As it is perfect gas we can use
(P₁V₁)/T₁=(P₂V₂)/T₂
Since this constant volume so
P₁/T₁=P₂/T₂
T₂ is change in temperature
T₂=1.00+273.16
T₂=274.16 K
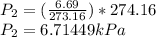
ΔP=6.71449-6.69
ΔP=0.0245 kPa
(b) As
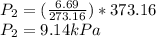
(c) Same steps as in part (a)
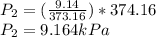
ΔP=9.164-9.14
ΔP=0.0245kPa