Answer:
0,051g of O₂ are consumed
Step-by-step explanation:
The reaction of precipitation of Fe(II) is:
4Fe(OH)⁺(aq) + 4OH⁻(aq) + O₂(g) + 2H₂O(l) → 4Fe(OH)₃(s)
The moles of Fe(II) you have in 85ml of 0.075 M Fe(II) are:
0,085L×0,075M = 6,4x10⁻³ moles of Fe(II)
For the reaction, you require 4 moles of Fe(II) and 1 mol of O₂(g) to precipitate the iron. Thus, moles of O₂(g) you require are:
6,4x10⁻³ moles of Fe(II)×
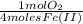 = 1,6x10⁻³ moles of O₂(g)
= 1,6x10⁻³ moles of O₂(g)
These moles are:
1,6x10⁻³ moles of O₂(g)×
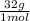 = 0,051g of O₂ are consumed
= 0,051g of O₂ are consumed
I hope it helps!