Answer: The binding energy of the given nucleus is 7.88 MeV/nucleon
Step-by-step explanation:
Nucleons are defined as the sub-atomic particles which are present in the nucleus of an atom. Nucleons are protons and neutrons.
We are given a nucleus having representation:
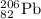
Number of protons = 82
Number of neutrons = 206 - 82 = 124
To calculate the mass defect of the nucleus, we use the equation:
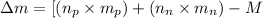
where,
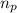 = number of protons = 82
= number of protons = 82
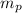 = mass of one proton = 1.007825 amu
= mass of one proton = 1.007825 amu
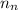 = number of neutrons = 124
= number of neutrons = 124
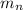 = mass of one neutron = 1.008665 amu
= mass of one neutron = 1.008665 amu
M = nuclear mass = 205.974440 amu
Putting values in above equation, we get:
![\Delta m=[(82* 1.007825)+(124* 1.008665)]-205.974440\\\\\Delta m=1.74167amu](https://img.qammunity.org/2020/formulas/chemistry/college/jh143m1rv8icgulqubepozwpxlzivr84o6.png)
To calculate the binding energy of the nucleus, we use the equation:
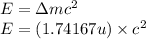
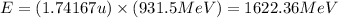 (Conversion factor:
(Conversion factor:
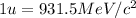 )
)
Number of nucleons in
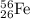 atom = 206
atom = 206
To calculate the binding energy per nucleon, we divide the binding energy by the number of nucleons, we get:
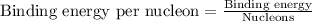
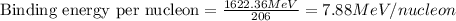
Hence, the binding energy of the given nucleus is 7.88 MeV/nucleon