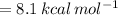Answer:
The activation energy is
Step-by-step explanation:
The gas phase reaction is as follows.
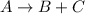
The rate law of the reaction is as follows.
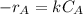
The reaction is carried out first in the plug flow reactor with feed as pure reactant.
From the given,
Volume "V" =
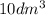
Temperature "T" = 300 K
Volumetric flow rate of the reaction
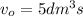
Conversion of the reaction "X" = 0.8
The rate constant of the reaction can be calculate by the following formua.
![V= (v_(0))/(k)[(1+\epsilon )ln((1)/(1-X)-\epsilon X)]](https://img.qammunity.org/2020/formulas/chemistry/college/sbls58wc9tdu4wmd0x5l9q22pjm8ds9vuv.png)
Rearrange the formula is as follows.
![k= (v_(0))/(V)[(1+\epsilon )ln((1)/(1-X)-\epsilon X)]............(1)](https://img.qammunity.org/2020/formulas/chemistry/college/6qis9lmw5k3nh4fyjk2ki2vdhgrnwwf4f7.png)
The feed has Pure A, mole fraction of A in feed
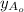 is 1.
is 1.
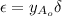
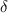 = change in total number of moles per mole of A reacte.
= change in total number of moles per mole of A reacte.
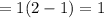
Substitute the all given values in equation (1)
![k=(5m^(3)/s)/(10dm^(3))[(1+1)ln (1)/(1-0.8)-1 * 0.8] = 1.2s^(-1)](https://img.qammunity.org/2020/formulas/chemistry/college/a2s5pjurlzx82gokxa0z3r2oi53lf06gs5.png)
Therefore, the rate constant in case of the plug flow reacor at 300K is
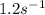
The rate constant in case of the CSTR can be calculated by using the formula.
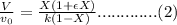
The feed has 50% A and 50% inerts.
Hence, the mole fraction of A in feed
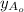 is 0.5
is 0.5
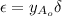
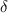 = change in total number of moles per mole of A reacted.
= change in total number of moles per mole of A reacted.
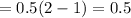
Substitute the all values in formula (2)
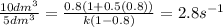
Therefore, the rate constant in case of CSTR comes out to be
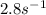
The activation energy of the reaction can be calculated by using formula
![k(T_(2))=k(T_(1))exp[(E)/(R)((1)/(T_(1))-(1)/(T_(2)))]](https://img.qammunity.org/2020/formulas/chemistry/college/43o87rhszll7iu1zy8w5hfhlqkzoohyuez.png)
In the above reaction rate constant at the two different temperatures.
Rearrange the above formula is as follows.
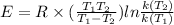
Substitute the all values.
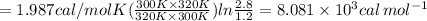
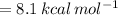
Therefore, the activation energy is