Answer:
Final pressure of the gas remaining in the first container is 3.5 atm
Step-by-step explanation:
Since it is given that temperature is constant , we can apply -
PV=constant , where
P = Pressure of the gas
V = Volume of the container in which the gas is contained
Initially,
For container 1 -
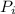 = 4 atm
= 4 atm
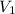 = 6 L
= 6 L
Finally,
For container 2 -
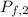 = 3 atm
= 3 atm
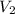 = 1 L
= 1 L
For container 1 -
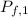 = ?
= ?
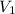 = 6 L
= 6 L
∴
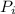
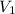 =
=
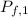
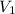 +
+
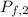
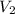
∴ 4×6 = (
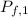 ×6) +(3×1)
×6) +(3×1)
∴
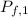 = 3.5 atm
= 3.5 atm