Answer: Positive, positive
Step-by-step explanation:
Entropy is the measure of randomness or disorder of a system. If a system moves from an ordered arrangement to a disordered arrangement, the entropy is said to decrease and vice versa.
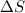 is positive when randomness increases and
is positive when randomness increases and
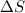 is negative when randomness decreases.
is negative when randomness decreases.
Exothermic reactions are defined as the reactions in which energy is released in the form of heat and
 for the reaction comes out to be negative.
for the reaction comes out to be negative.
Endothermic reactions are defined as the reactions in which energy is absorbed in the form of heat and
 for the reaction comes out to be positive.
for the reaction comes out to be positive.
For a cold pack, the energy has to be absorbed for the dissolution of ammonium nitrate in water so that the surroundings feel cold.Thus as energy is absorbed,
 for the reaction comes out to be positive.
for the reaction comes out to be positive.
The solid form of ammonium nitrate is getting converted to ions and thus entropy increases and thus
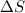 for the reaction comes out to be positive.
for the reaction comes out to be positive.