Answer:
The number of sulfur atoms in 1 g of sulfur is:-
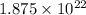 atoms
atoms
Step-by-step explanation:
Avogadro’s number represent the number of the constituent particles which are present in one mole of the substance. It is named after scientist Amedeo Avogadro and is denoted by
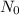 .
.
Avogadro constant:-
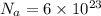
Given atomic mass of sulfur = 32 u
Which means that:-
32 g of sulfur contains
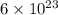 of atoms
of atoms
Also,
1 g of sulfur contains
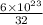 of atoms
of atoms
So, The number of sulfur atoms in 1 g of sulfur is:-
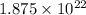 atoms
atoms