Answer:Frequency
Step-by-step explanation:
Photoelectric effect is described as the phenomenon of emission of electrons when light falls on the metal surface.
The theory assumes particle theory of light.
When photons hit the metal surface,the energy of photon is used to overcome to binding force between electron and nucleus.
The energy of photon is given by
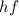 where
where
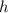 is placks constant and
is placks constant and
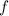 is frequency.
is frequency.
When the frequency of photon is low,the energy of photon may not be sufficient to emit a electron.
So,emission requires a minimum frequency.