Answer:
The minimum concentration of sodium sulfate required to producer precipitation is
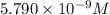 .
.
Step-by-step explanation:
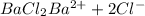
Concentration of barium chloride=
![[BaCl_2]=0.019 M](https://img.qammunity.org/2020/formulas/chemistry/college/l463i5rsqjd6ikalgm2dpk7rnnhqau3zkx.png)
Concentration of barium ions =
![[Ba^(2+)]](https://img.qammunity.org/2020/formulas/chemistry/college/nues6tz4ohyof39us2k87lfr5mfa7gnhu5.png)
1 mol of barium chloride gives 1 mol of barium ions.
![[Ba^(2+)]=[BaCl_2]=0.019 M](https://img.qammunity.org/2020/formulas/chemistry/college/e8g0dix374jxqqg49qdr1xln0knm51vzd5.png)
The solubility product for barium sulfate=
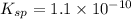
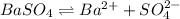
0.019 M S
The expression of solubility product for barium sulfate:
![K_(sp)=[Ba^(2+)]* S](https://img.qammunity.org/2020/formulas/chemistry/college/c84oew0zd1afhydpaxocp2cqbzqre64v5c.png)
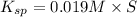
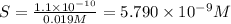
The minimum concentration of sulfate ions =
![[SO_4^(2-)]=5.790* 10^(-9) M](https://img.qammunity.org/2020/formulas/chemistry/college/95a0khhc7tpyak7jtktnvxuonky06f71lk.png)
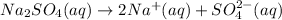
1 mole of sulfate ions are proceed form 1 mole of sodium sulfate solution.
Then
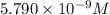 sulfate ions will be obtained from:
sulfate ions will be obtained from:
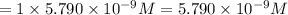 of sodium sulfate
of sodium sulfate
The minimum concentration of sodium sulfate required to producer precipitation is
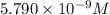 .
.