Answer: The volume of permanganate ion (potassium permanganate) is 10.0 mL
Step-by-step explanation:
To calculate the number of moles for given molarity, we use the equation:
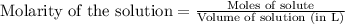 .....(1)
.....(1)
Molarity of ferrous sulfate solution = 0.381 M
Volume of solution = 20.0 mL = 0.020 L (Conversion factor: 1 L = 1000 mL)
Putting values in equation 1, we get:

For the given chemical equation:
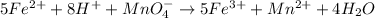
By Stoichiometry of the reaction:
5 moles of iron (II) ions (ferrous sulfate) reacts with 1 mole of permanganate ion (potassium permanganate)
So, 0.00762 moles of iron (II) ions (ferrous sulfate) will react with =
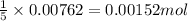 of permanganate ion (potassium permanganate)
of permanganate ion (potassium permanganate)
Now, calculating the volume of permanganate ion (potassium permanganate) by using equation 1, we get:
Molarity of permanganate ion (potassium permanganate) = 0.152 M
Moles of permanganate ion (potassium permanganate) = 0.00152 mol
Putting values in equation 1, we get:

Hence, the volume of permanganate ion (potassium permanganate) is 10.0 mL