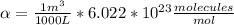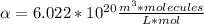Answer:
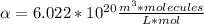
Step-by-step explanation:
Hi, the question states that there is a proportional relation between m (molarity) and n (number density), by following formula:
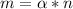
The units of alpha (
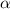 ) must help to balance the units of m and n.
) must help to balance the units of m and n.
1) First in both sides there are volume units liter and m3. So we need to express all volume in the same unit. Knowing that:
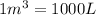
2) We also need to find a relation between mol and molecules. The relation is given by the Avogadro's number:
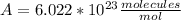
With this two numbers we can balance the units and find the value of
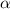 :
: