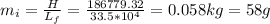Answer:
0.058 kg or 58 g
Step-by-step explanation:
The amount of heat energy required to cool 1.94 kg of water from 34 Celcius to 11 Celcius degrees is:
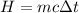
where m = 1.94 kg is the water mass, c = 4186 J/kg.C is water specific heat and
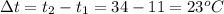
Hence H = 1.94 * 4186 * 23 = 186779.32 J
The required mass of ice to absorb this amount of heat would be