Answer:
B. 0.21 mol
General Formulas and Concepts:
Math
Pre-Algebra
Order of Operations: BPEMDAS
- Brackets
- Parenthesis
- Exponents
- Multiplication
- Division
- Addition
- Subtraction
Chemistry
Atomic Structure
- Using Dimensional Analysis
- Avogadro's Number - 6.022 × 10²³ atoms, molecules, formula units, etc.
Step-by-step explanation:
Step 1: Define
1.25 × 10²³ atoms Ca
Step 2: Identify Conversions
Avogadro's Number
Step 3: Convert
- Set up:
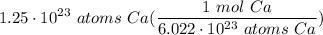
- Multiply:
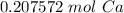
Step 4: Check
Follow sig fig rules and round. We are given 3 sig figs.
0.207572 mol Ca ≈ 0.208 mol Ca