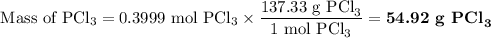Answer:
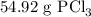
Step-by-step explanation:
We are given the masses of two reactants and asked to determine the mass of the product.
This looks like a limiting reactant problem.
1. Assemble the information
We will need a balanced equation with masses and molar masses, so let’s gather all the information in one place.
MM: 30.97 70.91 137.33
2P + 3Cl₂ ⟶ 2PCl₃
Mass/g: 12.39 42.54
2. Calculate the moles of each reactant
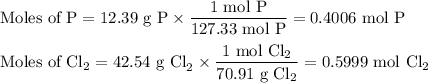
3. Calculate the moles of PCl₃ from each reactant
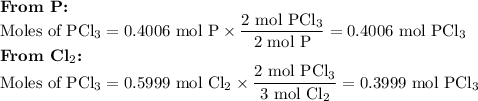
4. Identify the limiting reactant
Cl₂ is the limiting reactant because it gives fewer moles of PCl₃
5. Calculate the mass of PCl₃