Answer: The chemical equation is written below.
Step-by-step explanation:
Every balanced chemical equation follows law of conservation of mass.
This law states that mass can neither be created nor be destroyed but it can only be transformed from one form to another form. This also means that total number of individual atoms on reactant side must be equal to the total number of individual atoms on the product side.
The chemical equation for the reaction of elemental boron and oxygen gas follows:
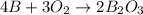
By Stoichiometry of the reaction:
4 moles of elemental boron reacts with 3 moles of oxygen gas to produce 2 moles of diboron trioxide.
The chemical equation for the reaction of diboron trioxide and water follows:
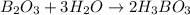
By Stoichiometry of the reaction:
1 mole of diboron trixoide reacts with 3 moles of water to produce 2 moles of boric acid.
Hence, the chemical equations are written above.