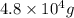Answer: The mass of molecular oxygen produced is
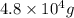
Step-by-step explanation:
To calculate the number of moles, we use the equation:
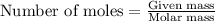 .....(1)
.....(1)
Given mass of ammonium perchlorate = 117.5 kg = 117500 g (Conversion factor: 1 kg = 1000 g)
Molar mass of ammonium perchlorate = 117.5 g/mol
Putting values in equation 1, we get:
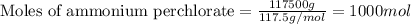
The chemical equation for the decomposition of ammonium perchlorate follows:
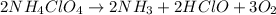
By Stoichiometry of the reaction:
2 moles of ammonium perchlorate produces 3 moles of molecular oxygen
So, 1000 moles of ammonium perchlorate will produce =
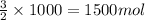 of molecular oxygen
of molecular oxygen
Now, calculating the mass of molecular oxygen by using equation 1, we get:
Molar mass of molecular oxygen = 32 g/mol
Moles of molecular oxygen = 1500 moles
Putting values in equation 1, we get:
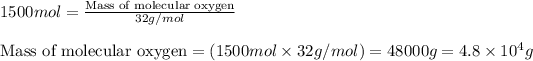
Hence, the mass of molecular oxygen produced is