Answer:
a) s = 8,1x10⁻³ mol/L
b) 29,7 g/100mL
c) 18,0 M
Step-by-step explanation:
For the equilibrium:
PbBr₂ ⇄ Pb²⁺ + 2 Br⁻
You can take the molar solubility of PbBr₂ (s) as:
[Pb²⁺] = s
[Br⁻] = 2s
As ksp is defined as:
ksp = [Pb²⁺] [Br⁻]²
Replacing:
2,1x10⁻⁶ = s * (2s)²
2,1x10⁻⁶ = 4s³
s = ∛2,1x10⁻⁶/4
s = 8,1x10⁻³ mol/L
b) The answer is g/100mL is:
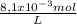 ×
×
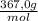 ×
×
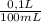 = 0,297 g/100mL
= 0,297 g/100mL
c) The concentration in mol/L is:
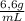 ×
×
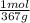 ×
×
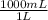 = 18,0 mol/L
= 18,0 mol/L
I hope it helps!