Answer: The volume of hydrogen gas produced is 4.24 L
Step-by-step explanation:
To calculate the number of moles, we use the equation:
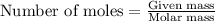
Given mass of aluminium = 3.12 g
Molar mass of aluminium = 26.98 g/mol
Putting values in above equation, we get:
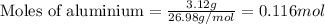
For the given chemical reaction:
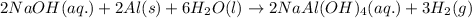
As, NaOH is present in excess. It is considered as an excess reagent.
Aluminium is the limiting reagent because it limits the formation of product.
By Stoichiometry of the reaction:
2 moles of aluminium produces 3 moles of hydrogen gas
So, 0.116 moles of aluminium will produce =
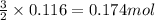 of hydrogen gas
of hydrogen gas
To calculate the volume of hydrogen gas produced, we use the ideal gas equation:
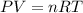
where,
P = pressure of the gas = 750 torr
V = Volume of the gas = ?
T = Temperature of the gas =
![20^oC=[20+273]K=293K](https://img.qammunity.org/2020/formulas/chemistry/high-school/2rqkt68ddzsgrhhdiygdxv8hg4exf1j7bf.png)
R = Gas constant =
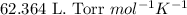
n = number of moles of the gas = 0.174 mol
Putting values in above equation, we get:
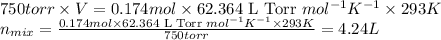
Hence, the volume of hydrogen gas produced is 4.24 L