Answer:
275 kPa
Step-by-step explanation:
mass of the gas=m=1.5 kg
initial volume if the gas=V₁=0.04 m³
initial pressure of the gas= P₁=550 kPa
as the condition is given final volume is double the initial volume
V₂=final volume
V₂=2 V₁
As the temperature is constant
T₁=T₂=T
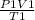 =
=
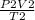
putting the values in the equation.
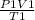 =
=
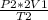
P₂=
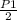
P₂=
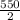
P₂=275 kPa
So the final pressure of the gas is 275 kPa.