Answer:
2,3 glasses of wine.
Step-by-step explanation:
In three hours, to produce a BAC of 0,10% for a 68kg person (5,4L blood) you need to consume:
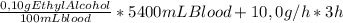 = 30,54g of ethyl alcohol
= 30,54g of ethyl alcohol
Assuming a 11,6% (v/v) of wine Where density of ethyl alcohol is 0,789 g/mL:
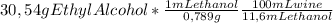 = 333,7 mL wine
= 333,7 mL wine
Thus, the person need to consume:
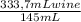 = 2,3 glasses of wine
= 2,3 glasses of wine