Answer:
equilibrium concentration is 2.477 ×
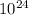 vacancies/m³
vacancies/m³
Step-by-step explanation:
given data
temperature = 600°C = 873 K
Energy of formation Al = 0.76 eV
atomic density = 2700 kg/m3
atomic weight of Al = 26.98 g/mol
to find out
equilibrium concentration of vacancies
solution
we have given atomic weight and density so we will apply formula that is
N =
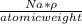
here ρ is density and Na is avogadro number i.e 6.022 ×
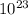 atoms/mol and N is related to density
atoms/mol and N is related to density
so
N =
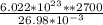
N = 6.027 ×
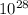 atoms/m³
atoms/m³
and
equilibrium concentration of vacancies is express as
n =
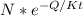
here t is temperature given and Q is energy formation and K is 8.62 ×
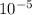 eV/atom
eV/atom
so
n =
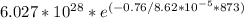
n = 2.477 ×
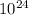 vacancies/m³
vacancies/m³
so equilibrium concentration is 2.477 ×
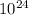 vacancies/m³
vacancies/m³