Answer:
Acid ammonium carbonate // Ammonium bicarbonate.
Step-by-step explanation:
Hello, a 4 is missing in the fist H, thus:
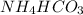
The presence of the hydrogen between the ammonium and the carbonates characterizes the salt as an acid salt, so you could name it as acid ammonium carbonate or ammonium bicarbonate (similar to the sodium bicarbonate which is
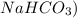 .
.
Best regards.