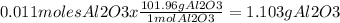Answer: The student should obtain 1.103 g of aluminum oxide
Step-by-step explanation:
- First we write down the equations that represent the aluminum hydroxide precipitation from the reaction between the aluminum nitrate and the sodium hydroxide:
Al(NO3)3 + 3NaOH → 3NaNO3 + Al(OH)3
Now, the equation that represents the decomposition of the hydroxide to aluminum oxide by heating it.
2Al(OH)3 → Al2O3 + 3H2O
- Second, we gather the information what we are going to use in our calculations.
Volumen of Al(NO3)3 = 40mL
Molar concentration of Al(NO3)3 = 0.541M
Molecular Weight Al2O3 = 101.96 g/mol
- Third, we start using the molar concentration of the aluminum nitrate and volume used to find out the total amount of moles that are reacting
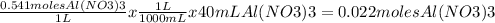
then we use the molar coefficients from the equations to discover the amount of Al2O3 moles produced
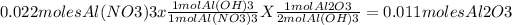
finally, we use the molecular weight of the Al2O3 to calculate the final mass produced.