Answer: The number of carbon and oxygen atoms in the given amount of carbon dioxide is
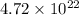 and
and
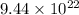 respectively
respectively
Step-by-step explanation:
To calculate the number of moles, we use the equation:
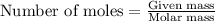
Given mass of carbon dioxide gas = 3.45 g
Molar mass of carbon dioxide gas = 44 g/mol
Putting values in above equation, we get:
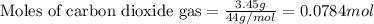
1 mole of carbon dioxide gas contains 1 mole of carbon and 2 moles of oxygen atoms.
According to mole concept:
1 mole of a compound contains
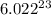 number of molecules
number of molecules
So, 0.0784 moles of carbon dioxide gas will contain
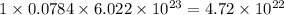 number of carbon atoms and
number of carbon atoms and
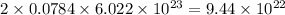 number of oxygen atoms
number of oxygen atoms
Hence, the number of carbon and oxygen atoms in the given amount of carbon dioxide is
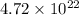 and
and
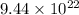 respectively
respectively