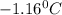Answer: Freezing point of a solution will be
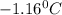
Step-by-step explanation:
Depression in freezing point is given by:
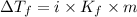
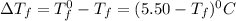 = Depression in freezing point
= Depression in freezing point
i= vant hoff factor = 1 (for non electrolyte)
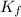 = freezing point constant =
= freezing point constant =
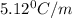
m= molality
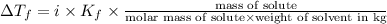
Weight of solvent (benzene)= 1480 g =1.48 kg
Molar mass of solute (octane) = 114.0 g/mol
Mass of solute (octane) = 220 g
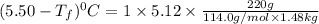

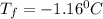
Thus the freezing point of a solution will be