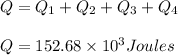Answer:
The amount of energy required is
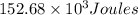
Step-by-step explanation:
The energy required to convert the ice to steam is the sum of:
1) Energy required to raise the temperature of the ice from -20 to 0 degree Celsius.
2) Latent heat required to convert the ice into water.
3) Energy required to raise the temperature of water from 0 degrees to 100 degrees
4) Latent heat required to convert the water at 100 degrees to steam.
The amount of energy required in each process is as under
1)
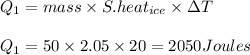
where
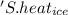 ' is specific heat of ice =
' is specific heat of ice =
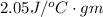
2) Amount of heat required in phase 2 equals
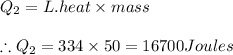
3) The amount of heat required to raise the temperature of water from 0 to 100 degrees centigrade equals
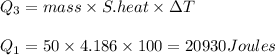
where
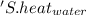 ' is specific heat of water=
' is specific heat of water=
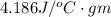
4) Amount of heat required in phase 4 equals
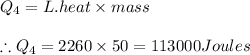
 Thus the total heat required equals
Thus the total heat required equals