Answer:
Part 1
(a) 0.0113
(b) 11300 ppm
(c) 1.82 *10⁻³
(d) 0.100 M
(e) 0.101 m
Part 2
(a) 9.45 *10⁻³
(b) mole fraction = 2.45 *10⁻³
(c) 11.3 ppm
Step-by-step explanation:
Chlorobenzene formula is C₆H₅Cl
Part 1: We are given a concentration of chlorobenzene in water of 100 mol/m³, and a density of the solution of 1.00 g/cm³.
(a) weight fraction C₆H₅Cl = mass C₆H₅Cl / mass solution
We know there are 100 moles of C₆H₅Cl per m³ of solution.
To get the mass of C₆H₅Cl we'll convert the moles to mass by using the molar mass:
Molar mass C₆H₅Cl = 6*12.011 + 5*1.00794 + 35.4527 = 112.558 g/mol
mass C₆H₅Cl = moles C₆H₅Cl * molar mass C₆H₅Cl
mass C₆H₅Cl = 100 moles * 112.558 g/mol = 11255.8 g
11255.8 g of C₆H₅Cl are in 1 m³ of solution.
Next we'll convert 1 m³ of solution to mass by using the density
mass solution = volume solution * density of solution
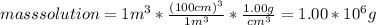
weight fraction C₆H₅Cl = 11256 g / 1.00 *10⁶ g = 0.0113
(b) ppm stands for "parts per million" and it is usually expressed as mg per Liter of solution
We already calculated that there are 11256 g or more exactly 11300 g of C₆H₅Cl in 1 m³ of solution, so lets convert to mg/L:
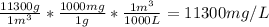
So the solution is 11300 ppm
(c) mole fraction = moles of C₆H₅Cl / total moles in solution
total moles = moles C₆H₅Cl + moles water
moles water = mass water / molar mass water
mass water = mass solution - mass C₆H₅Cl
moles of C₆H₅Cl = 100 moles
mass water = 1.00 *10⁶ g of solution - 11256 g = 988744 g of water
moles water = 988744 g / 18.0153 g/mol = 54884 moles water
total moles = 100 + 54884 = 54984 moles
mole fraction = 100 moles of C₆H₅Cl / 54984 moles = 1.82 *10⁻³
(d) Molarity = moles C₆H₅Cl / Liters of solution
We know the solution is 100 mol / m³ so we just have to convert the m³ to L:
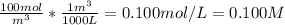
(e) Molality = moles C₆H₅Cl / kg water
We know that there are 100 moles per 988744 g of water, so we need to convert the grams of water to kilograms.
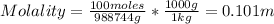
____________________________________
Part 2: Concentration of C₆H₅Cl in air is 0.100 mol/m³, at 25 °C and 1 atm.
Molar mass air = 28.84 g/mol
(a) weight fraction C₆H₅Cl = mass C₆H₅Cl / total mass
mass C₆H₅Cl = 0.100 mol * 112.558 g/mol = 11.26 g
total mass = mass C₆H₅Cl + mass air
mass air = moles air * molar mass air
moles air = total moles - moles C₆H₅Cl
We can calculate the total moles by using the ideal gas law:
P V = n R T
where P is pressure in atm, V is volume in L, n is the number of moles, R is the gas constant and T is temperature in Kelvin.
n = P V / R T
P = 1 atm
V = 1 m³ = 1000 L
R = 0.08206 L atm K⁻¹ mol⁻¹
T = 25 + 273.15 = 298 K
n = (1 atm * 1000 L) / (0.08206 L atm K⁻¹ mol⁻¹ * 298 K) = 40.89 moles
moles air = 40.89 - 0.100 = 40.79 moles air
mass air = 40.79 mol * 28.84 g/mol = 1176.4 g
total mass = 1176.4 g + 11.26 g = 1188 g
weight fraction = 11.26 g / 1188 g = 9.45 *10⁻³
(b) mole fraction = moles C₆H₅Cl / total moles
mole fraction = 0.100 / 40.89 = 2.45 *10⁻³
(c) ppm = mg C₆H₅Cl / Liters
We already know there are 11.26 g C₆H₅Cl in 1 m³, which is the same as 1000 L, so:
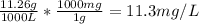
The concentration is 11.3 ppm