Answer: The average atomic mass of copper is 63.55 amu.
Step-by-step explanation:
Average atomic mass of an element is defined as the sum of masses of each isotope each multiplied by their natural fractional abundance.
Formula used to calculate average atomic mass follows:
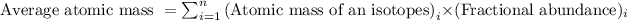 .....(1)
.....(1)
- For
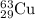 isotope:
isotope:
Mass of
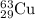 isotope = 62.94 amu
isotope = 62.94 amu
Percentage abundance of
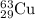 = 69.17 %
= 69.17 %
Fractional abundance of
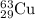 isotope = 0.6917
isotope = 0.6917
- For
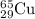 isotope:
isotope:
Mass of
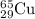 isotope = 64.93 amu
isotope = 64.93 amu
Percentage abundance of
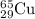 = 30.83 %
= 30.83 %
Fractional abundance of
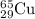 isotope = 0.3083
isotope = 0.3083
Putting values in equation 1, we get:
![\text{Average atomic mass of Copper}=[(62.94* 0.6917)+(64.93* 0.3083)]\\\\\text{Average atomic mass of copper}=63.55amu](https://img.qammunity.org/2020/formulas/chemistry/college/p3acwsai5wtw2tjpvm5ws0lzf67vl9c06m.png)
Hence, the average atomic mass of copper is 63.55 amu.