Answer: 64.1 ml of concentrated
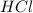 is taken and (250-64.4)= 185.6 ml f water is added to make 250.00 mL of 3.00 M HCl .
is taken and (250-64.4)= 185.6 ml f water is added to make 250.00 mL of 3.00 M HCl .
Explanation:
Given : 36 g of
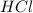 is dissolved in 100 g of solution.
is dissolved in 100 g of solution.
Density of solution = 1.18 g/ml
Thus volume of solution =
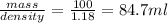
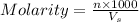
where,
n= moles of solute
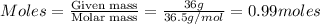
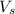 = volume of solution
= volume of solution
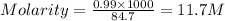
According to the dilution law,
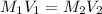
where,
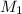 = molarity of stock
= molarity of stock
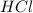 solution = 11.7M
solution = 11.7M
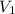 = volume of stock
= volume of stock
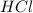 solution = V ml
solution = V ml
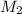 = molarity of diluted
= molarity of diluted
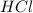 solution = 3.00 M
solution = 3.00 M
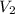 = volume of diluted
= volume of diluted
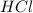 solution = 250.0 ml
solution = 250.0 ml
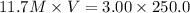
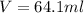
Thus 64.1 ml of stock
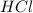 is taken and (250-64.4)= 185.6 ml of water is added to make 250.00 mL of 3.00 M HCl .
is taken and (250-64.4)= 185.6 ml of water is added to make 250.00 mL of 3.00 M HCl .