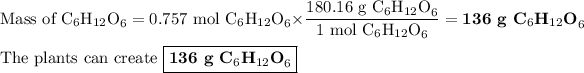Answer:
Step-by-step explanation:
We will need an equation with masses and molar masses, so let’s gather all the information in one place.
M_r: 44.01 180.16
6CO₂ + 6H₂O ⟶ C₆H₁₂O₆ + 6O₂
m/g: 200
1. Moles of CO₂
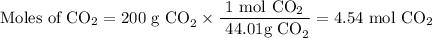
2. Moles of C₆H₁₂O₆
The molar ratio is 1 mol C₆H₁₂O₆:6 mol CO₂
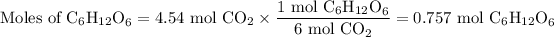
3. Mass of C₆H₁₂O₆