Step-by-step explanation:
As per the Hess’s law of constant heat summation, the heat absorbed or evolved in a given chemical equation is the same whether the process occurs in one step or several steps.
Hence, according to this law the chemical equation can be treated as ordinary algebraic expression and can be added or subtracted to yield the required equation. This means that the enthalpy change of the overall reaction is the sum of the enthalpy changes of the intermediate reactions.
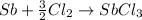
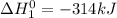 ..........(1)
..........(1)
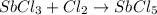
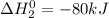 ..............(2)
..............(2)
The final reaction is as follows:
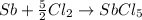
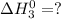 .............(3)
.............(3)
Therefore, adding (1) and (2) we get the final equation (3) and value of
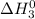 at 298 K will be as follows.
at 298 K will be as follows.
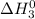 =
=
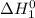 +
+
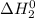
= -314 kJ + (-80) kJ
= -394 kJ
Thus, we can conclude that
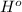 at 298 K for the given process is -394 kJ.
at 298 K for the given process is -394 kJ.