Answer: The concentration of evaporated solution is 0.794 M
Step-by-step explanation:
To calculate the molarity of the diluted solution, we use the equation:
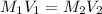
where,
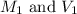 are the molarity and volume of the evaporated solution
are the molarity and volume of the evaporated solution
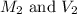 are the molarity and volume of initial solution
are the molarity and volume of initial solution
We are given:
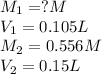
Putting values in above equation, we get:
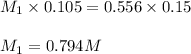
Hence, the concentration of evaporated solution is 0.794 M